Soft and hard materials exhibit mismatched behaviours, such as those in chemical or biochemical reactivity, mechanical response and environmental adaptability. Mitigating or leveraging these mismatches can yield interfacial chemical processes that are difficult to achieve, or inapplicable, in pure soft or pure hard phases. Exploration of interfacial mismatches and their associated chemical processes may reveal numerous opportunities in both fundamental studies and applications, in a manner similar to that of semiconductor heterojunctions and their contribution to solid-state physics and the semiconductor industry over the past few decades.
A) Dynamic instabilities in semiconductor-based materials growth
Reaction-diffusion and stick-slip motions are dynamic instabilities typically found in soft materials, and they have been used to model certain aspects of biological activities. However, they have rarely been explored or discovered in semiconductor materials growth. Our lab used silicon nanowire as a model system in which the catalyst, a silicon/gold alloy droplet, was considered to be the soft component in the study of the analogies of these two dynamic instabilities.
-
Deposition-diffusion-incorporation process for anisotropic silicon spicule
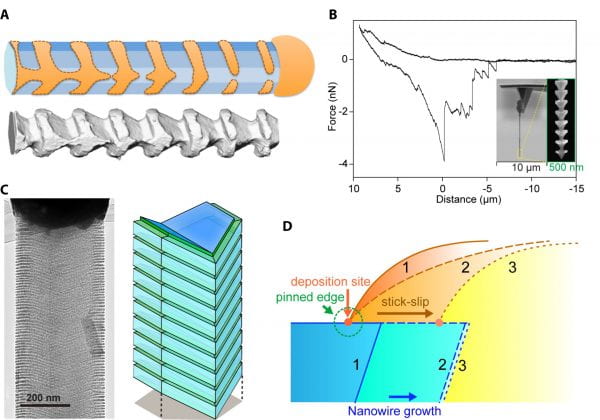
Metal can diffuse along semiconductor surfaces, and even trace amounts can change the surface behaviour. We exploited this phenomenon for three-dimensional (3D) mesoscale lithography by showing one example where iterated deposition-diffusion-incorporation of gold over silicon nanowires forms etchant-resistant patterns. This process is facet selective, producing mesostructured silicon spicules with skeleton-like morphology, tectonic motifs, and reduced symmetries (Fig. 1A). Atom-probe tomography, coupled with other quantitative measurements, indicates the existence and the role of individual gold atoms in forming 3D lithographic resists, a fundamentally different resist or mask mechanism in lithography. By carrying out extensive atomic force microscopy (AFM) studies, we also showed that the anisotropic silicon spicules have significantly enhanced mechanical interactions with extracellular biomaterials (Fig. 1B). It is important to note that current approaches to improve integration and signal transduction between medical devices and biological systems typically include conjugation of adhesive biomolecules on device surfaces and implementation of advanced device geometry and mechanics designs. Our work, therefore, took a completely different approach to achieving tight biointegration. The anisotropy-enhanced biointerfaces may be potentially utilized for new bioelectronics or electrophysiology device design and implementation.
-
Stick-slip process for porous silicon nanowire
Recently, we demonstrated that a liquid Au-Si alloy established in classical vapour-liquid-solid (VLS) growth can deposit ordered and 3D rings of isolated Au atoms over Si nanowire sidewalls (Fig. 1C). We revealed an atomic version of metal-assisted chemical etching (MACE), where single Au atoms can catalyze the etching of Si to create < 5 nm features. Experimental verification of this catalytic process in silicon nanowires yielded dopant-dependent, massive and ordered 3D grooves with spacing down to ~ 5 nm. We also discovered alloy droplet instability during the VLS growth, something which would promote a stick-slip motion along the nanowire sidewalls (Fig. 1D). Finally, using new atom patterning and atom-catalyzed etching, we revealed several complex crystal growth dynamics that were previously difficult to probe. In particular, the previous alloy droplet instabilities in VLS systems are all related to a configuration where the liquid is supported by the nanowire end facets. In this work, the new droplet instability is relevant to droplet wetting over the nanowire sidewall. This wetting configuration has received very little attention in the past, although theoretical analysis has predicted its existence. With regard to potential applications, atom-based self-labelling may be used to reveal the growth mechanisms for many other complex nanowire structures, such as epitaxial lateral nanowires and island-chain nanowires. This labelling may be applied broadly to chemistry and materials science with a role analogous to the use of fluorescent molecules in probing biological dynamics. Additionally, the parallel atom deposition and etching over nanowire sidewalls may be extended to atom-based catalytic lithography over planar substrates.
Figure 1: Dynamic instability. (A) A gold deposition-diffusion process for chemical resist that yields silicon spicule (lower panel). (B) A representative force-distance measurement from a Si spicule-based AFM probe. (C) The stick-slip motion of Si/Au alloy droplet can deposit periodic atomic Au lines for catalytic formation of porous Si nanowires. (D) Schematic diagram of the droplet stick-slip motion.
B) 3D calcite heterostructures for dynamic and deformable mineralized matrices
Naturally occurring minerals, which are part of highly dynamic ecosystems, have not been included in these studies. For example, calcite can be synthesized under ambient conditions, and it serves as a model system for understanding natural biomineralization. However, over the past two decades, most efforts using synthetic calcite have focused on mechanistic studies of crystal growth and kinetics, with only steady-state applications in very few areas (such as optics).
Recently, our lab demonstrated a new 3D calcite building block in which each structural unit has different growth kinetics and functions. Individual calcite crystals exhibit asymmetrical dumbbell shapes and are prepared by a parallel tectonic approach under ambient conditions. The silicone matrix immobilizes the epitaxial nucleation sites through self-templated cavities, which enables symmetry breaking in reaction dynamics and scalable manipulation of the mineral ensembles. Our lab expanded the applications for minerals, in particular exploring their utility in flexible electronics and underwater adhesives. Notably, the underwater adhesive involves a novel solid-state localized adhesion mechanism. Additionally, our lab synthesized a curved, monolithic and minimally-strained calcite lattice, with a morphology reminiscent of cocolithophore. All the growth processes, mechanistic studies and application demonstrations were performed under ambient conditions (e.g., at room temperature). Our work suggests a new pathway for dynamic inorganic systems by enabling regeneration sites.
C) Laser-assisted synthesis of silicon carbide for biointerfaces
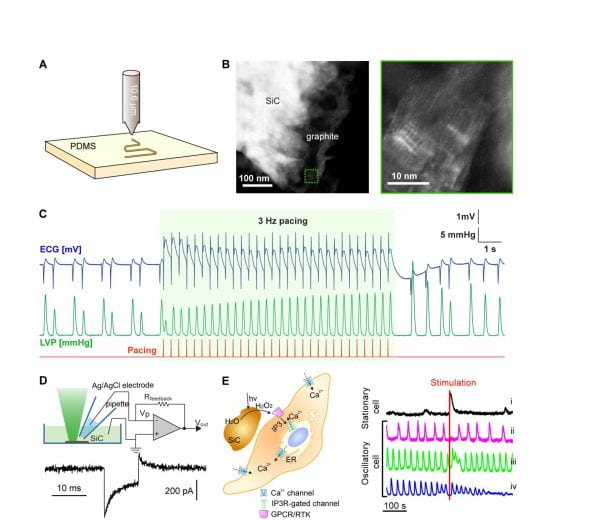
Laser-assisted processes are employed in material synthesis due to their ease of use, low cost, and unique capacity to generate complex phases. Laser-produced composites can expand the available design principles for materials and devices aimed at biological sensing or modulation. Recently, the Tian lab developed a laser-assisted fabrication method to prepare silicon carbide (SiC) from elastomer substrates (e.g., polydimethylsiloxane) for biomodulation (Fig. 2A-B). SiC is much more stable under physiological conditions than silicon, suggesting its potential incorporation in long-term biointerface studies.
The laser-assisted process incorporates nitrogen as an n-type dopant in SiC. Together with a supporting graphite network, the SiC in the flexible composite shows pseudocapacitive electrochemical and photoelectrochemical behaviours capable of modulating activity in isolated hearts (Fig. 2C) and cultured cells (Fig. 2D-E). In one example, we studied the calcium signals of individual cells in an ensemble of smooth muscle cells following photoelectrochemical stimulation from cell-interfaced SiC. We classified cells in the region of stimulation as stationary or oscillatory. The perturbations in calcium responses were consistent with experimental and theoretical studies of the behaviour of smooth muscle following an increase in inositol triphosphate (IP3) levels (likely due to H2O2-induced activation of G-protein coupled receptors or receptor tyrosine kinases). We also integrated our laser-produced SiC into a cell culture platform. Using a printed trench-like SiC device, we stimulated a sheet of smooth muscle cells. Our results revealed an overall increase in calcium levels and its propagation into adjacent cells as a calcium wave. Laser-assisted processes are employed in material synthesis due to their ease of use, low cost, and unique capacity to generate complex phases. Laser-produced composites can expand the available design principles for materials and devices aimed at biological sensing or modulation. Recently, the Tian lab developed a laser-assisted fabrication method to prepare silicon carbide (SiC) from elastomer substrates (e.g., polydimethylsiloxane) for biomodulation (Fig. 2A-B). SiC is much more stable under physiological conditions than silicon, suggesting its potential incorporation in long-term biointerface studies.
Our work on the laser-assisted fabrication of SiC-based devices suggests that laser writing can efficiently produce flexible and multifunctional semiconductor/elastomer composites for stable biointerface studies.
Figure 2. SiC for biointerfaces. (A) Schematics of the laser-assisted synthesis. (B) STEM images of the SiC/graphite composites. (C) Electrochemical pacing of isolated hearts using the SiC-based flexible electronics. (D-E) Photoelectrochemical properties of the SiC can elicit calcium responses in smooth muscle cells.
D) Micelle-assisted synthesis of nanoporous carbon for bioelectrical interfaces
Carbon nanostructures have been long studied as bioelectronics components due to their excellent electrical and mechanical properties and past successes in energy research. Nevertheless, the fabrication of cost-effective and safe carbon-based bioelectronics will require advanced synthesis methods.
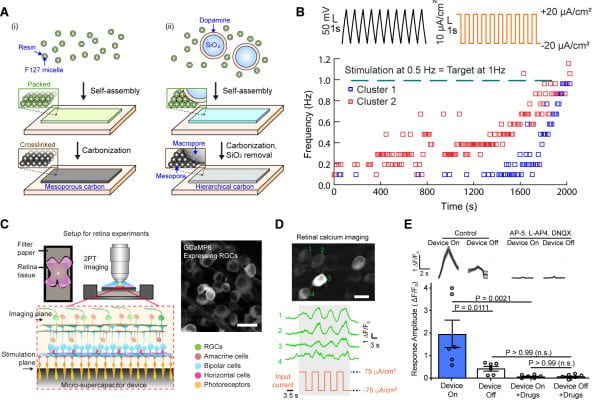
Recently, the Tian lab developed a synthesis method for hierarchical meso- and macro-porous carbon membranes using micelle-assisted self-assembly (Fig. 3A). We applied this method to fabricate binder-free carbon-based microelectrode arrays, using flexible SU-8 as a substrate. The hierarchical material design allows us to optimize its mechanical compliance and to leverage its biocompatible electrochemical properties for efficient biological stimulations. Inspired by micro-supercapacitors used in energy research, we explored the application of a comb-like interdigitated electrode design for in vitro stimulation and training of cells. For example, we applied a confined electric field to a cardiomyocyte monolayer and achieved efficient overdrive pacing and subthreshold levels of training in vitro (Fig. 3B). We also explored the stimulation of retina and heart ex vivo and stimulation of a sciatic nerve in vivo, demonstrating efficient coupling between the carbon nanostructures and excitable tissues (Fig. 3C-E). Our results illustrate how porous carbon materials may be utilized in biological modulation and how traditional energy research tools can be adapted to design bioelectronic systems.
Figure 3. Micelle-assisted self-assembly of nanoporous carbon for bioelectrical interfaces. (A) Schematics of the material preparation. (B) Subthreshold training of cardiomyocytes with the micro-supercapacitor-like device in vitro. (C-E) Modulation of retinal activities with super-capacitor-like device ex vivo.
E) Soft-hard interaction framework
Unique processes at the soft-hard interfaces are determined by the diverse soft material interactions (SS), the hard material interactions (HH), and the interactions between the soft and hard components (SH). Depending on the context of the discussion, the SS / HH could be the intramolecular interactions that hold the constituent phase together, the restoring forces upon elastic deformation, or physical or biological behaviors of the constituent. Likewise, the SH could be adhesion or bonding, chemical or biological recognition, or signal conversion (e.g., from photoelectrochemical to electrophysiological signals) at the soft-hard interfaces.
Based on the relative dominance of the roles played by the three interactions in determining the spatial organization or the temporal sequence of the soft-hard interface, we propose a soft-hard ‘interaction framework’ which covers both the spatial and temporal aspects of the interactions (Fig. 4, Video 1).
Each color domain in Fig. 4A-4D (spatial aspect) is the overlapped area from two half-triangles which are defined by two inequalities that need to be satisfied simultaneously. The arrows in Fig. 4E and 4F (temporal aspect) denote the signal transduction directions. This framework, especially the notion of ‘relative dominance’ is primarily qualitative, although quantitative information such as bending stiffness or power can inform the material choices for interface designs. For example, the bending stiffness of a brain tissue could be in the range of 10-4 ~ 10-1 nN×m. If we would like to establish very compliant brain interfaces (i.e., SS > HH) from pure silicon-based membranes, we could use a 10 nm-thick membrane (bending stiffness, ~ 10-5 nN×m). Another example is establishing a biointerface between a single bacterium and a single silicon nanowire for optically triggered metabolic control. The power consumption of a bacterial cell is ~ 10-12 W, while the power output from a coaxial silicon nanowire-based photovoltaic device is ~ 10-10-10-9 W. This power difference makes the signal transduction from silicon nanowire to bacterium possible, which has been proved experimentally.
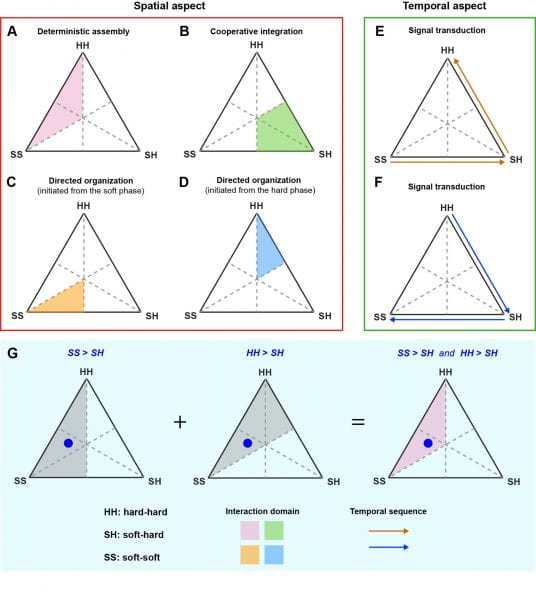
Figure 4. We propose a new “Interaction Framework” at the soft-hard interfaces. The interactions can be (bio)chemical, mechanical, electrical, etc. (A-D) represent the spatial aspect, while (E-F) represent the temporal aspect of the interaction framework. Depending on the specific target/domain of a system, different interaction frameworks can co-exist in a single system. (G) Similar to the interpretation of a conventional phase diagram, the relative distance from a point (blue dot) to the three corners of the framework triangle suggests the relative dominance of the roles of SS, SH, and HH interactions; the shorter the distance to one corner, the more dominant the interaction that is represented by the corner. Each color domains in A–D are the overlapped area from two half-triangles (in grey) as two inequalities need to be satisfied simultaneously. The approach to derive the domain for the case of “deterministic assembly” is given in G.
Video 1. Description of the soft-hard interaction framework.
Selected Publications
- Z. Q. Luo, Y. W. Jiang, B. D. Myers, D. Isheim, J. S. Wu, J. F. Zimmerman, Z. A. Wang, Q. Q. Li, Y. C. Wang, X. Q. Chen, V. P. Dravid, D. N. Seidman and B. Z. Tian, Atomic gold-enabled three-dimensional lithography for silicon mesostructures, Science, 2015, 348, 1451-1455. Link
- J. Yi, Y. C. Wang, Y. W. Jiang, I.-W. Jung, W. J. Liu, V. De Andrade, R. Q. Xu, R. Parameswaran, I. R. Peters, R. Divan, X. H. Xiao, T. Sun, Y. Lee, W.-I. Park, B. Z. Tian, 3D calcite heterostructures for dynamic and deformable mineralized matrices, Nature Communications, 2017, 8, article number: 509. Link
- Y. Fang, Y. W. Jiang, M. J. Cherukara, F. Y. Shi, K. Koehler, G. Freyermuth, D. Isheim, B. Narayanan, A. W. Nicholls, D. N. Seidman, S. K. R. S. Sankaranarayanan, B. Z. Tian, Alloy-assisted deposition of three-dimensional arrays of atomic gold catalyst for crystal growth studies, Nature Communications, 2017, 8, article number: 2014. Link
- V. Nair, J. Yi, D. Isheim, M. Rotenberg, L. Y. Meng, F. Y. Shi, X. Q. Chen, X. Gao, A. Prominski, Y. W. Jiang, J. P. Yue, C. T. Gallagher, D. N. Seidman, B. Z. Tian, Laser writing of nitrogen-doped silicon carbide for biological modulation. Science Advances, 2020, 6 (34), eaaz2743. Link
- Y. Fang, A. Prominski, M. Y. Rotenberg, L. Y. Meng, H. A. Ledesma, Y. Y. Lv, J. P. Yue, E. Schaumann, J. Jeong, N. Yamamoto, Y. W. Jiang, B. Elbaz, W. Wei, B. Z. Tian, Micelle-enabled self-assembly of porous and monolithic carbon membranes for bioelectronic interfaces, Nature Nanotechnology, 2021. DOI: 10.1038/s41565-020-00805-z Link
- Y. Fang, X. Yang, Y. L. Lin, J. Y. Shi, A. Prominski, C. Clayton, E. Ostroff, B. Z. Tian. Dissecting biological and synthetic soft-hard interfaces for tissue-like systems, Chemical Reviews, 2021, 10.1021/acs.chemrev.1c00365, and the video. Link