Our team is at the forefront of developing living bioelectrical interfaces, and we believe these innovations hold immense potential for the future. We are dedicated to pushing the boundaries of what is possible in this exciting field, with the ultimate goal of improving human health and advancing our knowledge of living systems.
A) Macroporous bioelectronics scaffold for organ-on-a-chip — “Cyborg tissue”
In our pioneering study, we explored the innovative domain of integrating nanoelectronics with synthetic biomaterials, leading to the development of macroporous nanowire nanoelectronic scaffolds (nanoES). These scaffolds, characterized by their 3D macroporous structure, flexibility, and biocompatibility, are designed to mimic natural tissue scaffolds (Fig. 1). We employed silicon nanowire field-effect transistors (FETs) within these scaffolds, which enabled us to record both extracellular and intracellular signals with subcellular precision. This groundbreaking approach allows for a minimally invasive 3D interface of electronics with cells in tissue, overcoming the limitations of previous planar devices. Our nanoES are distinct in their ability to combine with both synthetic and natural biomaterials, providing an electronic sensory function to extracellular matrices (ECMs) and a suitable biochemical environment for tissue culture. The development of these nanoES involved innovative design and fabrication techniques, utilizing electron beam lithography and photolithography to create structures that replicate the submicron features of ECMs.
Furthermore, we conducted extensive evaluations to confirm the functionality and utility of our nanoES in various biological contexts. We successfully integrated the nanoES with a range of biomaterials, such as collagen, alginate, and poly(lactic-co-glycolic acid) (PLGA), creating hybrid scaffolds that maintained the intrinsic characteristics of the nanowire FET devices. These hybrid nanoES were tested in 3D cultures with different cell types, demonstrating minimal impact on cell viability, and thus are suitable for in vitro studies like drug screening assays. Remarkably, our research showed the potential of nanoES in biomedical applications, notably in 3D pH sensing, which can be pivotal for detecting changes in physiological conditions such as inflammation or ischemia. These findings represent a significant stride in the convergence of nanoelectronics and biological systems, paving the way for advanced synthetic tissues and novel biomedical devices.

Figure 1: We developed macroporous, flexible, and free-standing bioelectronic scaffolds, along with their hybrids with synthetic or natural biomaterials. These bioelectronic scaffolds have been successfully used for 3D cultures and enable integrated sensory capabilities, such as real-time monitoring of electrical activity, drug responses in tissue models, and pH changes in vascular smooth muscle constructs.
B) Cellular-scale biointegrated photostimulation — “Cyborg cell”
We have developed a novel cell-silicon hybrid system that seamlessly combines the natural behaviors of living cells with the advanced functionalities of silicon nanotechnology. Our innovative approach, distinct from conventional bioelectronics, utilizes cardiac myofibroblasts integrated with silicon nanowires (SiNWs) to achieve interconnect-free cellular modulation with exceptional subcellular precision. This hybrid system is particularly applied in cardiac systems, where it has significantly advanced our ability to synchronize the electrical activity of cardiomyocytes (Fig. 2). This enables an in-depth exploration of heterocellular bioelectric coupling, both in vitro and in vivo. Our method stands out for its minimally invasive nature and its ability to integrate seamlessly with cardiac tissues, thus avoiding the fibrotic responses commonly associated with bare SiNWs. Through our research, we have uncovered critical insights into the mechanisms of electrical coupling and calcium flux propagation, offering new perspectives on cardiac physiology and pathophysiology.
Building on this foundation, we envision a similar approach for the neural system, where we could employ oligodendrocytes hybridized with internal nanowires to establish intercellular interfaces with neurons. This concept could enable the photostimulation of neurons through oligodendrocyte/nanowire hybrids, thereby paving the way for revolutionary advances in neurological research and treatment. We anticipate that this neural system application would mirror the high precision and minimal invasiveness of our cardiac system approach. The potential of this technology to manipulate neural activities and its implications for neuronal connectomics and neurological disorder treatments are vast and promising. Our work not only marks a significant leap in bioelectronic interfaces, particularly in cardiac research, but also opens a multitude of possibilities for probing and understanding the intricacies of biological systems in both clinical and research contexts.
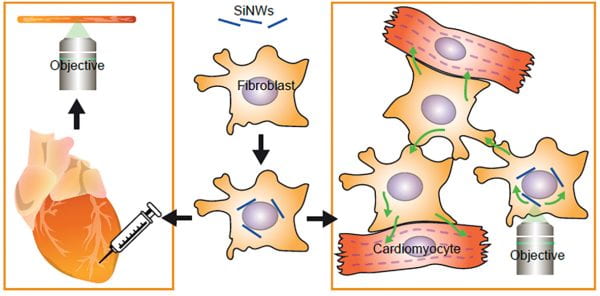
Figure 2: Our research introduces a novel cell-silicon hybrid system, integrating cardiac myofibroblasts with silicon nanowires (SiNWs), which offers a less invasive, interconnect-free approach for precise modulation of cellular activities, particularly in cardiac systems. This method, distinct from traditional bioelectronics, synchronizes cardiomyocyte electrical activities and provides vital insights into cardiac bioelectric coupling and calcium flux mechanisms.
C) Biomineralization-enabled photoelectrochemical biosynthesis
We explored an innovative method of periplasmic biomineralization within Gram-negative bacteria. This novel approach led to the formation of semiconductor nanoclusters, possessing unique fluorescent properties, within the bacteria’s periplasmic space. A striking aspect of these nanoclusters is their metastable, disordered, and defect-rich nature, which, quite unexpectedly, contributes to increased levels of adenosine triphosphate (ATP) and enhanced malate production. Additionally, we demonstrated the critical role of cysteine in facilitating this biomineralization process, enabling the bacteria to tolerate and process high concentrations of heavy metals such as Cd2+, Pb2+, and Hg2+. This finding is particularly significant as it showcases the potential for environmental remediation through the efficient removal of toxic heavy metals.
Our research further looks into the complexities of the biomineralization pathway, identifying the production of hydrogen sulfide (H2S) from cysteine as a key component. This H2S-mediated pathway is essential for the precipitation of these nanoclusters. We also discovered the system’s capability to handle multiple metal ions simultaneously, forming high-entropy sulfide aggregates that are suitable for a wide range of applications. Another remarkable aspect of our work is the development of a living bioreactor concept, which utilizes bacteria and filter membranes for scalable and continuous biomineralization processes. Lastly, we highlighted the potential for semi-artificial photosynthesis through optically modulated biointerfaces (Fig. 3). Here, the biomineralized nanoclusters play a pivotal role in increasing ATP production and boosting the biosynthesis of valuable chemicals such as malate under light stimulation. These findings mark significant strides in biotechnology, especially in sustainable environmental remediation and energy production.
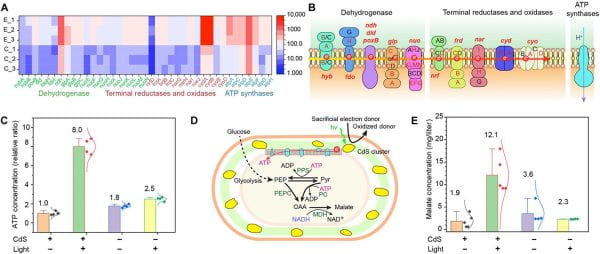
Figure 3: We developed a method for periplasmic biomineralization in Gram-negative bacteria, leading to the formation of fluorescent, metastable semiconductor nanoclusters that enhance ATP and malate production, while enabling efficient heavy metal removal. This breakthrough, involving H2S-mediated pathways and a living bioreactor system, paves the way for applications in environmental remediation and semi-artificial photosynthesis, underscoring significant advancements in sustainable biotechnology.
D) Living bioelectrical interfaces for drug-free and adaptive healthcare
We are developing living bioelectrical interfaces that move beyond passive sensing toward active, biological participation in healthcare. Rather than treating tissues as inert substrates for electronics, our approach integrates soft bioelectronics with living components—such as cells or microbes—embedded within tissue-like hydrogel interfaces. This strategy allows electronic systems to communicate with biology not only through electrical signals, but also through biochemical and immunological pathways.
By designing interfaces that are mechanically compliant, biologically active, and electrically functional, we enable long-term, stable coupling between devices and living tissues. These living interfaces can sense physiological states, respond to environmental changes, and actively modulate local tissue microenvironments. Importantly, they open a path to drug-free therapies, where disease states are regulated through electrical cues, biological signaling, and host–microbe interactions rather than systemic pharmaceuticals.
We apply this framework to inflammatory and immune-related conditions, where localized and adaptive modulation is critical. Living bioelectrical interfaces allow us to monitor disease progression in real time while simultaneously guiding tissue recovery through controlled bioelectrical and biogenic interactions. Built with sustainability and scalability in mind, these systems rely on soft materials, low-power electronics, and renewable biological components.
Overall, our work establishes living bioelectrical interfaces as a new paradigm for healthcare—one that leverages cooperation between electronics and living systems to achieve precise, adaptive, and sustainable therapeutic outcomes.

Figure 4: Living bioelectronics with a bioactive interface for wireless skin monitoring and therapy. (A) This schematic shows the overall idea of living bioelectronics: the device can continuously collect information from the skin, help identify disease states, and deliver therapy in a single, integrated system. (B) The function of the living biointerface arises from the combined roles of electronics, soft hydrogel, and living bacteria. The biopolymer matrix keeps the bacteria alive, while the bacteria interact with and regulate the skin’s immune environment. At the same time, the electronics record electrical signals from the skin and apply controlled electrical stimulation to maintain safety. The hydrogel stores the bacteria, keeps them viable over time, conforms gently to the skin, and adheres securely, allowing stable and long-term signal collection. (C) Compared with traditional bioelectronic interfaces, ABLE brings together biological activity, soft tissue-like mechanics, and electrical function in a single platform. (D) Photographs show several physical designs of the ABLE device, highlighting its small size and flexible form. (E) Photographs illustrate how the ABLE interface makes direct, conformal contact with the skin, enabling stable bioelectrical interaction.
Selected References
- B. Z. Tian*, J. Liu*, T. Dvir*, L. H. Jin, J. H. Tsui, Q. Qing, Z. G. Suo, R. Langer, D. S. Kohane and C. M. Lieber, Macroporous nanowire nanoelectronic scaffolds for synthetic tissues, Nature Mater., 2012, 11, 986-994. Link
- J. F. Zimmerman, R. Parameswaran, G. Murray, Y. Wang, M. Burke, B. Z. Tian. Cellular uptake and dynamics of unlabeled freestanding silicon nanowires. Science Advances, 2016, 2:e1601039. Link
- M. Y. Rotenberg, N. Yamamoto, E. N. Schaumann, L. Matino, F. Santoro, B. Z. Tian, Living myofibroblast-silicon composites for probing electrical coupling in cardiac systems. PNAS, 2019, 116, 22531-22539. Link
- Y. L. Lin*, J. Y. Shi*, W. Feng*, J. P. Yue, Y. Q. Luo, S. Chen, B. Yang, Y. W. Jiang, H. C. Hu, C. K. Zhou, F. Y. Shi, A. Prominski, D. V. Talapin, W. Xiong, X. Gao, B. Z. Tian. Periplasmic biomineralization for semi-artificial photosynthesis. Science Advances, 2023, DOI: 10.1126/sciadv.adg5858. Link
- J. Y. Shi*, S. Kim*, P. J. Li, F. Y. Dong, C. W. Yang, B. Nam, C. Han, E. Eig, L. L. Shi, S. M. Niu, J. P. Yue, B. Z. Tian, Active biointegrated living electronics for managing inflammation. Science, 2024, DOI: 10.1126/science.adl1102. Link
Go to: General Research
